
PDF Publication Title:
Text from PDF Page: 079
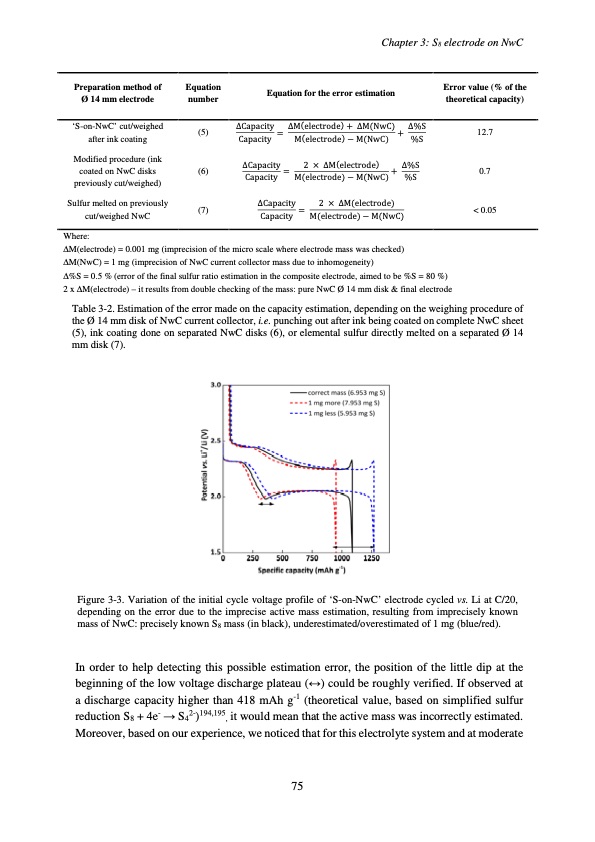
‘S-on-NwC’ cut/weighed (5) after ink coating Sulfur melted on previously (7) cut/weighed NwC ∆Capacity = ∆M(electrode) + ∆M(NwC) + ∆%S Capacity M(electrode) − M(NwC) %S ∆Capacity = 2 × ∆M(electrode) Capacity M(electrode) − M(NwC) 12.7 < 0.05 Chapter 3: S8 electrode on NwC Preparation method of Ø 14 mm electrode Equation number Equation for the error estimation Error value (% of the theoretical capacity) Modified procedure (ink coated on NwC disks previously cut/weighed) (6) ∆Capacity = 2 × ∆M(electrode) + ∆%S Capacity M(electrode) − M(NwC) %S 0.7 Where: ∆M(electrode) = 0.001 mg (imprecision of the micro scale where electrode mass was checked) ∆M(NwC) = 1 mg (imprecision of NwC current collector mass due to inhomogeneity) ∆%S = 0.5 % (error of the final sulfur ratio estimation in the composite electrode, aimed to be %S = 80 %) 2 x ∆M(electrode) – it results from double checking of the mass: pure NwC Ø 14 mm disk & final electrode Table 3-2. Estimation of the error made on the capacity estimation, depending on the weighing procedure of the Ø 14 mm disk of NwC current collector, i.e. punching out after ink being coated on complete NwC sheet (5), ink coating done on separated NwC disks (6), or elemental sulfur directly melted on a separated Ø 14 mm disk (7). Figure 3-3. Variation of the initial cycle voltage profile of ‘S-on-NwC’ electrode cycled vs. Li at C/20, depending on the error due to the imprecise active mass estimation, resulting from imprecisely known mass of NwC: precisely known S8 mass (in black), underestimated/overestimated of 1 mg (blue/red). In order to help detecting this possible estimation error, the position of the little dip at the beginning of the low voltage discharge plateau (↔) could be roughly verified. If observed at a discharge capacity higher than 418 mAh g-1 (theoretical value, based on simplified sulfur reduction S8 + 4e- → S42-)194,195, it would mean that the active mass was incorrectly estimated. Moreover, based on our experience, we noticed that for this electrolyte system and at moderate 75PDF Image | Accumulateur Lithium Soufre
PDF Search Title:
Accumulateur Lithium SoufreOriginal File Name Searched:
WALUS_2015_archivage.pdfDIY PDF Search: Google It | Yahoo | Bing
Sulfur Deposition on Carbon Nanofibers using Supercritical CO2 Sulfur Deposition on Carbon Nanofibers using Supercritical CO2. Gamma sulfur also known as mother of pearl sulfur and nacreous sulfur... More Info
CO2 Organic Rankine Cycle Experimenter Platform The supercritical CO2 phase change system is both a heat pump and organic rankine cycle which can be used for those purposes and as a supercritical extractor for advanced subcritical and supercritical extraction technology. Uses include producing nanoparticles, precious metal CO2 extraction, lithium battery recycling, and other applications... More Info
| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |