
PDF Publication Title:
Text from PDF Page: 030
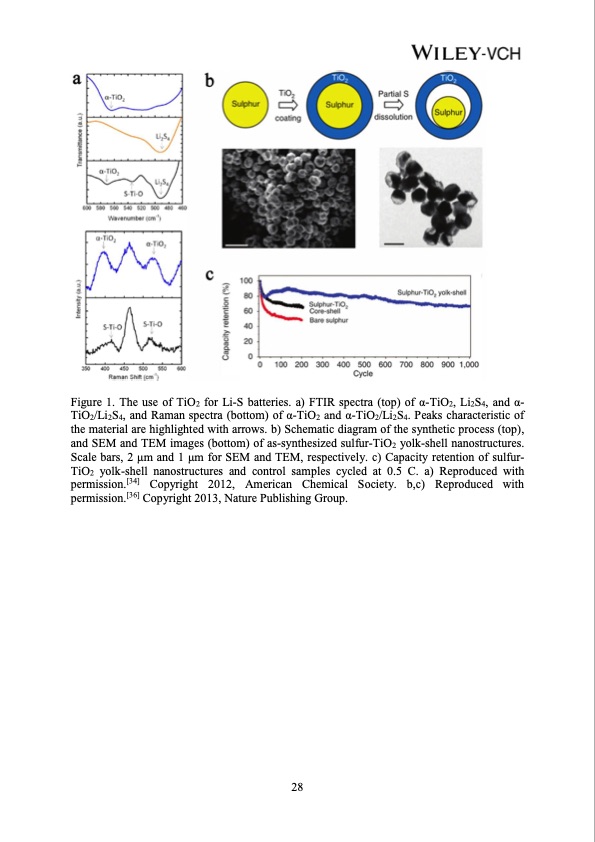
Figure 1. The use of TiO2 for Li-S batteries. a) FTIR spectra (top) of α-TiO2, Li2S4, and α- TiO2/Li2S4, and Raman spectra (bottom) of α-TiO2 and α-TiO2/Li2S4. Peaks characteristic of the material are highlighted with arrows. b) Schematic diagram of the synthetic process (top), and SEM and TEM images (bottom) of as-synthesized sulfur-TiO2 yolk-shell nanostructures. Scale bars, 2 μm and 1 μm for SEM and TEM, respectively. c) Capacity retention of sulfur- TiO2 yolk-shell nanostructures and control samples cycled at 0.5 C. a) Reproduced with permission.[34] Copyright 2012, American Chemical Society. b,c) Reproduced with permission.[36] Copyright 2013, Nature Publishing Group. 28PDF Image | Advances in Polar Materials for Lithium-Sulfur Batteries
PDF Search Title:
Advances in Polar Materials for Lithium-Sulfur BatteriesOriginal File Name Searched:
212718644.pdfDIY PDF Search: Google It | Yahoo | Bing
Sulfur Deposition on Carbon Nanofibers using Supercritical CO2 Sulfur Deposition on Carbon Nanofibers using Supercritical CO2. Gamma sulfur also known as mother of pearl sulfur and nacreous sulfur... More Info
CO2 Organic Rankine Cycle Experimenter Platform The supercritical CO2 phase change system is both a heat pump and organic rankine cycle which can be used for those purposes and as a supercritical extractor for advanced subcritical and supercritical extraction technology. Uses include producing nanoparticles, precious metal CO2 extraction, lithium battery recycling, and other applications... More Info
| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |