
PDF Publication Title:
Text from PDF Page: 036
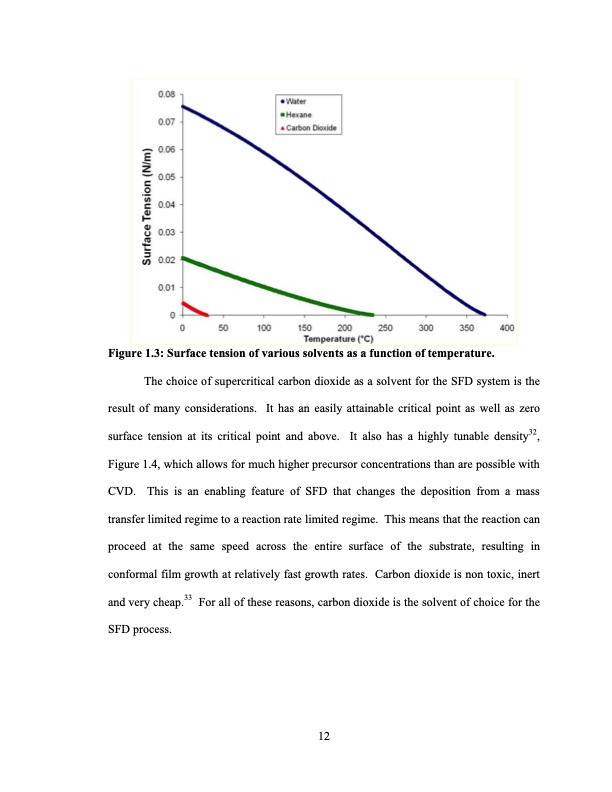
Figure 1.3: Surface tension of various solvents as a function of temperature. The choice of supercritical carbon dioxide as a solvent for the SFD system is the result of many considerations. It has an easily attainable critical point as well as zero surface tension at its critical point and above. It also has a highly tunable density32, Figure 1.4, which allows for much higher precursor concentrations than are possible with CVD. This is an enabling feature of SFD that changes the deposition from a mass transfer limited regime to a reaction rate limited regime. This means that the reaction can proceed at the same speed across the entire surface of the substrate, resulting in conformal film growth at relatively fast growth rates. Carbon dioxide is non toxic, inert and very cheap.33 For all of these reasons, carbon dioxide is the solvent of choice for the SFD process. 12PDF Image | Supercritical Fluid Deposition Of Thin Metal Films
PDF Search Title:
Supercritical Fluid Deposition Of Thin Metal FilmsOriginal File Name Searched:
Supercritical-Fluid-Deposition-Of-Thin-Metal-Films-Kinetics-Mec.pdfDIY PDF Search: Google It | Yahoo | Bing
Sulfur Deposition on Carbon Nanofibers using Supercritical CO2 Sulfur Deposition on Carbon Nanofibers using Supercritical CO2. Gamma sulfur also known as mother of pearl sulfur and nacreous sulfur... More Info
CO2 Organic Rankine Cycle Experimenter Platform The supercritical CO2 phase change system is both a heat pump and organic rankine cycle which can be used for those purposes and as a supercritical extractor for advanced subcritical and supercritical extraction technology. Uses include producing nanoparticles, precious metal CO2 extraction, lithium battery recycling, and other applications... More Info
| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |