
PDF Publication Title:
Text from PDF Page: 006
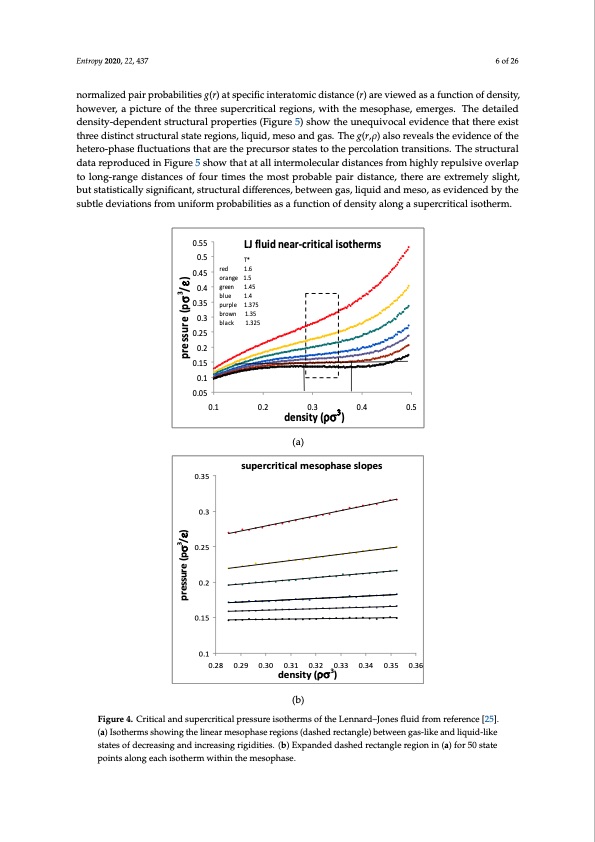
Entropy 2020, 22, 437 6 of 26 normalized pair probabilities g(r) at specific interatomic distance (r) are viewed as a function of density, however, a picture of the three supercritical regions, with the mesophase, emerges. The detailed density-dependent structural properties (Figure 5) show the unequivocal evidence that there exist three distinct structural state regions, liquid, meso and gas. The g(r,ρ) also reveals the evidence of the hetero-phase fluctuations that are the precursor states to the percolation transitions. The structural data reproduced in Figure 5 show that at all intermolecular distances from highly repulsive overlap to long-range distances of four times the most probable pair distance, there are extremely slight, but statistically significant, structural differences, between gas, liquid and meso, as evidenced by the subtle deviations from uniform probabilities as a function of density along a supercritical isotherm. (a) 6 of 27 0.55 0.5 0.45 0.4 0.35 0.3 0.25 0.2 0.15 0.1 0.05 LJ fluid near-critical isotherms T* red 1.6 orange 1.5 green 1.45 blue 1.4 purple 1.375 brown 1.35 black 1.325 0.1 0.2 0.3 0.4 0.5 density (ρσ3) 0.35 0.3 0.25 0.2 0.15 0.1 0.28 0.29 0.30 0.31 0.32 0.34 0.35 0.36 supercritical mesophase slopes 30.33 density (ρσ ) (b) FFigiguurere44..CCrritiitcicaallaannddssupeerrccrriittiiccallpressureisotherms of the Lennard–Jonesflfluiidffrromrreeffeerreennccee[2[255].]. (a(a)) Isothermsshsohwowinigngthethlienelairnmeaersomphesaosephreagsieonrse(gdiaosnhsed(dreacsthaendgler)ebcteatnwgelen) gbaest-wlikeenandgalisq-ulikide-liakned lisqtuatieds-loikfedestcareteasionfgdaencdreianscirnegasaindgriingcirdeiatiseisn.g(bri)gEidxiptaiensd.e(bd)dEaxsphaedndredctadnagslheerdegrieocntaing(lae)rfeogri5o0nsitnat(ea) fopro5in0tstaltoenpgoeinatcshailsonthgeeramchwiistohtihnetrhmewmietshoinphthaseem. esophase. Another advantage of computer simulation is that detailed structural data can be obtained that helps to explain the phenomenological behaviour at the molecular level. The Lennard–Jones fluid was found to have a critical isotherm at kTc/ε = 1.3365 ± 0.0005 (where k is Boltzmann’s constant and ε is the attractive minimum energy of the L-J pair potential). Several supercritical isotherms were studied in more detail with a very high precision, including the kT/ε = 1.5 isotherm over the whole density range. pressure (pσ3/ε) pressure (pσ3/ε)PDF Image | Supercritical Fluid Gaseous and Liquid States
PDF Search Title:
Supercritical Fluid Gaseous and Liquid StatesOriginal File Name Searched:
entropy-22-00437.pdfDIY PDF Search: Google It | Yahoo | Bing
Sulfur Deposition on Carbon Nanofibers using Supercritical CO2 Sulfur Deposition on Carbon Nanofibers using Supercritical CO2. Gamma sulfur also known as mother of pearl sulfur and nacreous sulfur... More Info
CO2 Organic Rankine Cycle Experimenter Platform The supercritical CO2 phase change system is both a heat pump and organic rankine cycle which can be used for those purposes and as a supercritical extractor for advanced subcritical and supercritical extraction technology. Uses include producing nanoparticles, precious metal CO2 extraction, lithium battery recycling, and other applications... More Info
| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |