
PDF Publication Title:
Text from PDF Page: 061
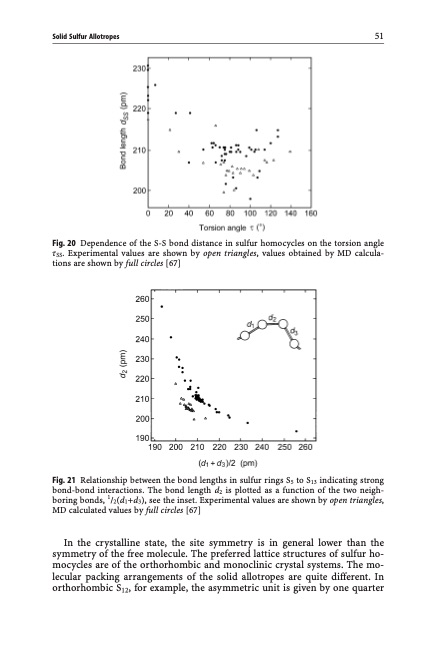
Solid Sulfur Allotropes 51 Fig. 20 Dependence of the S-S bond distance in sulfur homocycles on the torsion angle tSS. Experimental values are shown by open triangles, values obtained by MD calcula- tions are shown by full circles [67] Fig. 21 Relationship between the bond lengths in sulfur rings S5 to S13 indicating strong bond-bond interactions. The bond length d2 is plotted as a function of the two neigh- boring bonds, 1/2(d1+d3), see the inset. Experimental values are shown by open triangles, MD calculated values by full circles [67] In the crystalline state, the site symmetry is in general lower than the symmetry of the free molecule. The preferred lattice structures of sulfur ho- mocycles are of the orthorhombic and monoclinic crystal systems. The mo- lecular packing arrangements of the solid allotropes are quite different. In orthorhombic S12, for example, the asymmetric unit is given by one quarterPDF Image | Topics in Current Chemistry
PDF Search Title:
Topics in Current ChemistryOriginal File Name Searched:
Elemental-Sulfur-und-Sulfur-Rich-Compounds-I.pdfDIY PDF Search: Google It | Yahoo | Bing
Sulfur Deposition on Carbon Nanofibers using Supercritical CO2 Sulfur Deposition on Carbon Nanofibers using Supercritical CO2. Gamma sulfur also known as mother of pearl sulfur and nacreous sulfur... More Info
CO2 Organic Rankine Cycle Experimenter Platform The supercritical CO2 phase change system is both a heat pump and organic rankine cycle which can be used for those purposes and as a supercritical extractor for advanced subcritical and supercritical extraction technology. Uses include producing nanoparticles, precious metal CO2 extraction, lithium battery recycling, and other applications... More Info
| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |